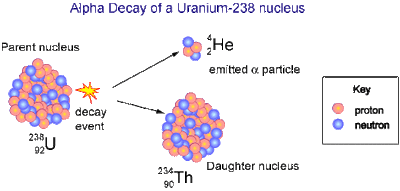
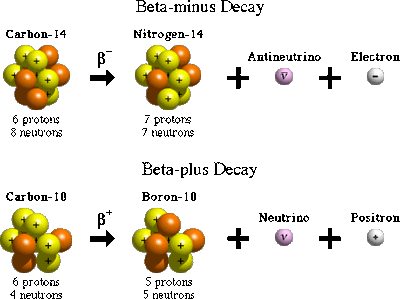Alpha decay-
Alpha decay is the process that an unstable atom can become more stable. During alpha decay, an atom's nucleus gives away two protons and two neutrons, this is what scientists call an alpha particle. Since an atom loses two protons during alpha decay, it changes from one element to another. Alpha decay only occurs in atoms with very heavy nuclei, this is usually where the mass number is greater than 100. Paper can stop Alpha decay.
For example radio isotope uranium-238 can undergo alpha decay.
Travel at a 10% speed of light
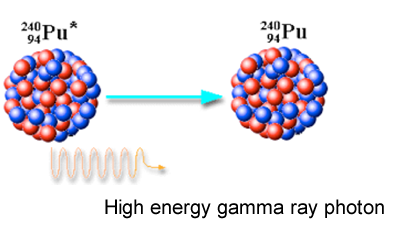
Gamma decay-
Gamma decay is the release of energy in the form of gamma rays. In gamma decay, a high energy photon is emitted from the nucleus to attain a lower energy configuration. Protons and neutrons rearrange inside of a nucleus, but do not emit a particle. Instead they emit a form of electromagnetic waves known a gamma decay. They are similar to X-rays but are way more powerful. A 2 inch slab of concrete or lead can stop Gamma decay. Gamma decays is not a particle, it's rays of energy.
Travel at a speed of light
Beta decay-
Beta decay occurs when the nucleus ejects a beta particle. Beta particles are identical to electrons and therefore are small and have a negative charge.When a nucleus undergoes beta decay, a neutron is converted into a proton. This increase the atomic number by 1 which therefore means it forms a new element. The mass does not change because the number of protons and neutrons stay the same amount. Aluminum can stop Beta decay.
For example carbon-14 atom has 6 protons and 8 neutrons. When the atom ejects a beta particle, one of the neutrons becomes a proton, this atom becomes nitrogen-14 with 7 protons and 7 neutrons.
Travels at a 90% speed of light.
Alpha decay is the process that an unstable atom can become more stable. During alpha decay, an atom's nucleus gives away two protons and two neutrons, this is what scientists call an alpha particle. Since an atom loses two protons during alpha decay, it changes from one element to another. Alpha decay only occurs in atoms with very heavy nuclei, this is usually where the mass number is greater than 100. Paper can stop Alpha decay.
For example radio isotope uranium-238 can undergo alpha decay.
Travel at a 10% speed of light
Gamma decay-
Gamma decay is the release of energy in the form of gamma rays. In gamma decay, a high energy photon is emitted from the nucleus to attain a lower energy configuration. Protons and neutrons rearrange inside of a nucleus, but do not emit a particle. Instead they emit a form of electromagnetic waves known a gamma decay. They are similar to X-rays but are way more powerful. A 2 inch slab of concrete or lead can stop Gamma decay. Gamma decays is not a particle, it's rays of energy.
Travel at a speed of light
Beta decay-
Beta decay occurs when the nucleus ejects a beta particle. Beta particles are identical to electrons and therefore are small and have a negative charge.When a nucleus undergoes beta decay, a neutron is converted into a proton. This increase the atomic number by 1 which therefore means it forms a new element. The mass does not change because the number of protons and neutrons stay the same amount. Aluminum can stop Beta decay.
For example carbon-14 atom has 6 protons and 8 neutrons. When the atom ejects a beta particle, one of the neutrons becomes a proton, this atom becomes nitrogen-14 with 7 protons and 7 neutrons.
Travels at a 90% speed of light.