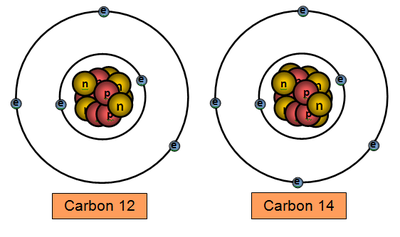
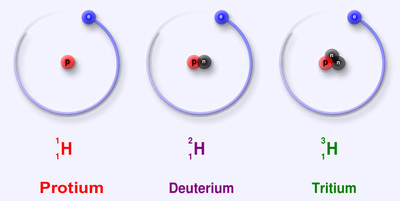
The general definition of an Isotope is atoms that have the same number of protons but different a number of neutrons.
An isotope is 2 or more atoms with the same amount of protons, but different amounts of neutrons. They are a differing form of a single element.
For example, Carbon 12 and Carbon 14 are both isotopes of carbon, one with 6 neutrons and one with 8 neutrons = both with 6 protons.
Isotopes of an element have the same atomic number but different atomic mass.